Randomized Controlled Trial: Targeted Neck Cooling in the Treatment of the Migraine Patient
Adam S Sprouse-Blum, MD, Alexandra K Gabriel, BSN, RN, Jon P Brown, BS, and Melvin HC Yee, MD
Abstract
Cold therapy has long been the number one self-care treatment employed for migraine without aura and the second most common for migraine with aura, yet its mechanism remains elusive. In this study, a mechanism by which this time-tested therapy works is proposed (by cooling the blood passing through intracranial vessels) in an attempt to further elucidate its beneficial effects. The study is designed as a randomized, controlled, crossover clinical trial utilizing an adjustable wrap containing two freezable ice packs targeting the carotid arteries at the neck, where they come close to the skin surface. Fifty-five participants successfully completed the study. Pain at onset, as recorded on a visual analog scale, was similar between the two treatment arms. Maximum pain reduction was observed at the 30 minute time point with a 31.8% ± 15.2% decrease in pain in the treatment arm compared to a 31.5% ± 20.0% increase in pain at the same time interval in the control arm. These findings confirm the application of a frozen neck wrap at onset of migraine headache targeting the carotid arteries at the neck significantly reduced recorded pain in participants with migraine headaches (P<.001).
Introduction
The treatment of migraine with cold therapy has been used for over 150 years. James Arnott was the first to document his application of salt and ice mixtures in the treatment of headache in 1849.1 Since then, many studies have re-examined this technique utilizing various methods of cold application. Ucler utilized a frozen gel cap that covered the entire head and included a chin strap,2 Robbins utilized an elastic head band with reusable gel packs centered over the forehead, 3 and Diamond utilized simple gel packs held by hand to the area of pain.4 Lance utilized a complicated helmet system combining cold with variable heat and pressure.5 In an interesting study by Friedman,6 hollow metal tubes chilled by circulating cold water were applied to the periapical area of the maxillary molars. All of the aforementioned studies reported symptomatic relief with their respective cold therapies. Today, many patients utilize cold therapy as part of their treatment regime and Zanchin reports that,4,7 of all self administered pain-relieving maneuvers, cold therapy is the most common maneuver applied in migraine without aura and the second most common maneuver applied in migraine with aura second only to compression.8
The physiologic effects of cryotherapy are well studied on many systems. Those systems most intimately involved with the application of cryotherapy to migraine are the vascular, neurologic and endocrine. With respect to the vascular system, cold induces vasoconstriction with subsequent decreased downstream blood flow.9–16 A secondary reactive vasodilation, referred to as the “Hunting response,”16–19 is thought to occur in less than 20 to 30 minutes of persistent cold application.20,18 However, the practical effects of this phenomenon have been drawn into question.21 In addition to vasoconstriction, cryotherapy also decreases local edema.10,14 This decrease in edema is thought to result from decreased vascular permeability which in turn is thought to be due to decreased release of inflammatory mediators.22, 23 In regards to the neurologic system, cryotherapy induces analgesia by slowing nerve conduction velocity with sensory fibers being affected before motor fibers.24–29 More specifically, small myelinated fibers are affected first followed by large myelinated fibers with unmyelinated fibers being affected last.29 In following with the gate control mechanism of pain,30 cryotherapy induces analgesia by affecting the small myelinated nociceptive pain afferents. As for the endocrine system, cryotherapy decreases metabolic and enzymatic activity.26, 31, 32 Under such conditions, there is a decreased tissue demand for ATP with a subsequent decrease in local oxygen demand.31, 33 Finally, a discussion of the physiologic effects of cryotherapy as they apply to the migraine patient would not be complete without acknowledging the important contribution of the placebo effect on patient’s subjective assessment of their pain.34,35
In this pilot study, a wrap containing frozen ice packs targeting the carotid arteries at the front of the neck is used to attempt to evaluate the benefits of targeted carotid cooling in the migraine patient. The study stems from a hypothesis that the mechanism through which cold therapy is effective in treating migraine is by cooling the blood passing through intracranial vessels. As such, cold applied to the head would have to penetrate the skull to reach the target vessels. Marathon runners apply ice packs to the armpits and groin after a race because this is where large blood vessels come close to the skin surface. This study uses a similar concept to address the carotid arteries where they come close to the skin surface at the neck to further elucidate the benefits of this time proven therapy.
Methods
This study is designed as a randomized, controlled, crossover clinical trial utilizing an adjustable neoprene neck wrap that holds two freezable ice packs targeting the carotid arteries in the treatment of migraine headaches. The study was approved by the institutional review boards of the three affiliated institutions. Participants were recruited from the general public through posters, local media, and physician referral from March through June of 2012 and followed through September, 2012. All participants were screened by the same investigator by means of a pre-research questionnaire to ensure they met the following inclusion criteria: male or female, 18–65 years old and met current International Headache Society (IHS) International Classification of Headache Disorders (ICHD-2) criteria for migraine (with or without aura) at time of entry into the study.36 Participants who were taking more than three medications (including over-the-counter) specifically for migraine at the time of interview, or had failed triptan therapy in the past, were excluded from the study. Participants were not monetarily compensated for their participation; however, they were allowed to keep the investigational neck wrap at the conclusion of the study. Eligible participants were randomized into one of two arms: frozen (treatment) or non-frozen/room temperature (control) according to when they entered the study (odd numbered participants started in the treatment arm and even numbered participants started in the control arm). Participants were educated that cold therapy is the most common self-care treatment employed in migraine, and that this study evaluates this method of treatment in a novel location, at the neck. A concerted effort was made not to lead participants as to what to expect from either arm. All participants were educated how to appropriately apply the neck wrap (Figure 1) with an emphasis on ensuring good skin contact at the anterior most portion of the neck…where the carotid arteries lie. The wrap itself was utilized in exactly the same manner regardless of which arm the participant was in. Specifically, the ice packs were maintained in the wrap during both phases of the study; in the treatment arm, participants were instructed to place the entire wrap (including ice packs) in the freezer. Participants were given freedom to store the wrap in whatever location they felt was most practical with emphasis given to being able to access the wrap at migraine onset. Of note, during the frozen (treatment) phase of the study, storage locations were somewhat limited due to the fact the wrap had to be kept in a freezer. Participants already utilizing pharmacologic migraine prophylaxis at study entry were allowed to continue taking these medications during the course of the study. However, they were instructed to utilize the wrap prior to taking all acute migraine medication(s). Data was recorded on a one page migraine pain diary using the visual analog scale (VAS) shown in Figure 2, at the following time intervals: onset, 15 minutes, 30 minutes, and one hour. The wrap was worn for the first 30 minutes of each episode, at which point the participant removed the wrap and waited another 30 minutes (with the wrap off) to record their final (one hour) pain score. Furthermore, participants were given the option to utilize their acute/rescue migraine medication(s) at any time after they recorded their 15 minute pain score. However, if they elected to do so, they were instructed to cease taking data from that point forward. In all trials, participants answered the following four YES/NO questions: “Do you feel the wrap helped you?” “If it DID work, did your headache come back?” “Did you experience any side-effects from neck wrap use? If YES, please list:” and “Did you have to take any medications to treat your headache?” At one month, participants were asked to cross over from one arm to the other (frozen went to non-frozen and vice-versa) for the second month. If a participant did not experience a migraine (and take data) during the first month, they were asked to wait until they recorded data from at least one episode before switching arms. A minimum of one migraine episode in each arm (frozen and non-frozen) was required to complete the study. All participants received monthly follow-up reminders by email and/or phone. Data was returned in a self-addressed envelope.
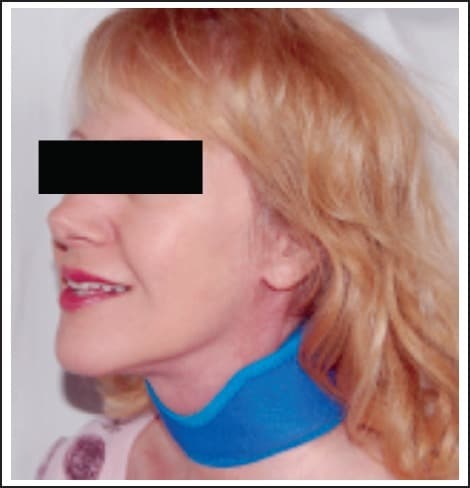
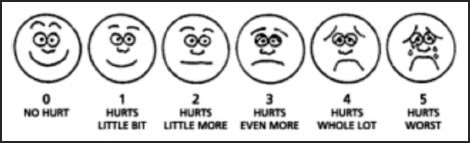
Figure 2. Visual analogue scale.
Mean pain scores were calculated across all participants at each time interval and compared to pain at onset to determine percent change from baseline. Subset analyses were used to further characterize percent change in those without and with aura. Fisher’s exact test with mid-p, two-sided values of P<0.05 was used to evaluate for statistical significance.
Results
Demographics
One hundred one participants met IHS ICHD-2 criteria for migraine and were enrolled in the study and 64 submitted data. Of these, 9 had incomplete or incorrectly filled out data forms. As such, 55 participants were included in the data analysis. Of these, 25.5% (n=14) met IHS criteria for migraine with aura, the rest (74.5%) met criteria for migraine without aura. 85.5% of participants (n=47) were female. Participant ages varied from 19 to 64 with a mean age of 43.1 ± 11.4 years. Reported frequency of migraine attacks varied from less than one per month to daily with a median frequency of 5.5 attacks per month and an interquartile range of 4.63 (25th percentile) and 28.75 (75th percentile). Of note, reported median frequency and interquartile range are approximations due to the fact that individual averages were used when participants reported a range in monthly migraine frequency.
Pain Score (using 0–5 visual analog scale shown in Figure 2)
Table 1 shows the mean pain scores at each time interval for both treatment and control arms. Mean pain score at migraine onset was similar in both arms (2.83 ± 0.26 frozen, compared to 2.61 ± 0.25 non-frozen). Participants in the treatment arm reported a maximum decrease in pain at 30 minutes, recording a 31.8% ± 15.2% decrease in pain over onset. Those without aura reported a decrease at 30 minutes of 39.3% ± 13.4% and those with aura a decrease of 11.2% ± 43.8%. Once the frozen neck wrap was removed, pain improvement fell to 27.3% ± 17.5% at one hour. In comparison, those in the control arm reported a 31.5% ± 20.0% increase in pain at 30 minutes, further increasing in severity to 35.4% ± 24.1% at one hour. The information presented in Table 1 is represented graphically in Figure 3.
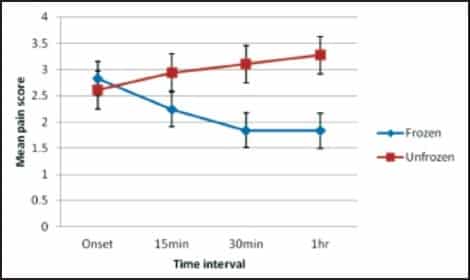
Figure 3: Mean pain score at each time interval.

Clinical Response
In response to the question: “Do you feel the wrap helped you?” 77.0% (n=55) of participants in the treatment arm responded “Yes” compared to 6.4% (n=55) in the control arm. In response to the question “If it DID work, did your headache come back?” 52.0% (n=50) responded “Yes” in the treatment arm compared to 20.0% (n=5) in the control arm. In response to the question “Did you experience any side-effects from neck wrap use?” 3 participants responded “Yes” in the treatment arm with 2 participants experiencing neck and/or shoulder tightness and 1 experiencing nausea with rapid heartbeat compared to 1 participant in the control arm who experienced mild dizziness. None of the aforementioned side-effects led to discontinuation of treatment. In response to the question “Did you have to take any medications to treat your headache?” 57.6% (n=55) responded “Yes” in the treatment arm compared to 83.6% (n=55) in the control arm. Response to this question was not limited to the 1 hour trial period only. That is, it included both participants who abandoned the experiment during the trial period, opting to utilize a rescue medication, and those who successfully completed the 1 hour trial but subsequently took medication due to incomplete relief or return of headache.
Discussion
In hypothesizing how the frozen neck wrap was significantly more effective at decreasing participant recorded pain score over control, effects on the following three systems are proposed: (1) vascular, (2) neurologic and (3) endocrine with its effects on the vascular system being most important. With respect to the vascular system, it is possible that the blood passing through the carotid arteries was cooled sufficiently to induce a physiologic decrease in release of local inflammatory mediators from the walls of the intracranial vessels with an associated decrease in vascular permeability and a decrease in local nociceptive stimulation (including that due to plasma extravasation). Also, there may have been a contribution of cold induced local physiologic vasoconstriction similar to that observed with triptan type medications, albeit through a different mechanism. Of note, the role of vasoconstriction in the treatment of migraine remains unclear. In regards to the neurologic system, a minimal contribution of analgesia associated with the small diameter myelinated fibers located around the cranial vessels is proposed. And as for the endocrine system, there may be a minimal contribution of decreased metabolic activity in locally cooled areas.
An interesting finding in this study is participants without aura recorded a greater mean decrease in pain score compared to participants with aura (39.3 ± 13.4% compared to 11.2 ± 43.8% at 30 minutes). While this association did not achieve statistical significance, it is in line with Zanchin’s observation that cold is the most commonly applied maneuver in migraine without aura and the second most common in migraine with aura. Its underpinnings may lie in the pathophysiologic consequences of cortical spreading, more closely associated with migraine with aura. More specifically, since the intervention is primarily targeting a vascular contribution of migraine, those who progressed to cortical spreading may have had a diminished response. For this reason, participants were instructed to apply the wrap at onset of migraine symptoms. However, some participants reported they were unable to do so because they woke up with migraine or did not have access to the intervention until after migraine onset.
Another interesting finding is the subjective return of migraine pain once the ice packs melted as reflected by the 52.0% of participants who answered “Yes” to the question: “If it DID work, did your headache come back?” in the treatment arm. Objectively, this association was not observed at the one hour time point with essentially stable mean pain scores at 30 minutes and one hour (1.84 ± 0.27 and 1.83 ± 0.33, respectively). However, participants responding to this question were not instructed to limit their response to the one hour study period. Rather, it was left to the participant’s discretion to evaluate whether they experienced return of migraine pain. While it would have been ideal to have a 2 or 4 hour pain score time point, this was not done in an attempt to keep the study design simple and as manageable as possible for participants. Future studies should include at least a 2 hour follow up to better evaluate for return of migraine pain once treatment is ceased.
The limitations of this study are numerous. However, significant attempts were made to mitigate as many of them as reasonable to create as clear a picture as possible. With respect to selection bias, participants were recruited from the general public through posters, local media, and physician referral. As such, it is possible that participants who elected to be in the study were more motivated than the average migraineur to actively treat their migraines, or suffered more frequent or severe attacks. Along these lines, participants were not monetarily compensated with the exception of allowing them to keep the investigational neck wrap at study conclusion, and the inclusion and exclusion criteria were tailored to attempt to accurately reflect the majority of migraineurs in the general population. Another source for selection bias suffered was a high attrition rate (46.5%). The majority of the observed attrition was due to loss of university students to summer break and graduation, incomplete or incorrectly filled out data, duration of study, and lack of migraine frequency. To minimize these losses, all participants received monthly follow-up by email and/or phone calls. Of note, participants who returned their completed data did not vary widely with those who did not, with the exception of a moderately better return rate in those with more frequent attacks, as expected. The small size of the study is an obvious limitation. Fortunately, statistical significance was achieved. Neck size and carotid anatomy is variable between participants. However, a velcro attachment system that allowed the wrap to be adjustable across a wide range of neck circumferences was utilized, and each participant was shown how to appropriately apply the wrap by the same investigator. Nevertheless, there is inherent variability in wrap application based on personal preference for tightness. A few participants found the wrap uncomfortable. The wrap was constructed of neoprene material and gave a scent that was bothersome to some participants. As such, participants were encouraged to remove the ice packs and wash the wrap prior to use. The silica based blue gel packs utilized in the study were relatively small and only remained frozen for approximately 15–20 minutes (multiple participants complained they wished the packs were larger and stayed colder longer). This relatively short duration was taken advantage of when designing the study to allow for the evaluation of return of migraine pain once the gel packs melted. Another important study limitation is that targeted neck cooling is intended for headaches with a vascular component (of which migraines were assumed to be included). Current IHS ICHD-2 criteria were used to screen participants prior to study enrollment. However, as with all diagnostic criteria, some participants who were false positive were included in the study and some false negatives were missed. While obvious, this is particularly true in a condition as diverse and poorly understood as migraine. As with all pain studies, one must consider the contribution of placebo effect on the outcomes. This study was designed as a cross-over study such that each participant served as her or his own control to help deal with the contribution of placebo to perceived efficacy. This is not a perfect design, however. Although emphasis was placed on not leading the patient as to what to expect from either condition, it was likely obvious to many, which was the treatment arm. Hróbjartsson identified non-blinded assessments as a major limitation in randomized clinical trial that involve subjective measurement scales.37 That being said, even with the best designed studies, participants commonly suspect whether they are in the treatment group and only rarely can true blinding exist. Given this caveat, we hope our data is found statistically relevant to the point of intriguing the research and clinical communities to challenge our results and to further elucidate the benefits of targeted neck cooling in the treatment of the migraine patient.
Conclusion
The application of a frozen neck wrap at onset of migraine targeting the carotid arteries at the neck significantly reduced recorded pain in participants with migraine headaches. Future studies to further elucidate the mechanism by which targeted carotid cooling alleviates migraine include utilizing a cold water circulator connected to a neck garment such that temperature and duration of treatment can be accurately controlled. This would allow investigators to identify whether a dose-dependent relationship exists. Alternatively, a variety of imaging modalities could be used to identify downstream neurologic and/or vascular changes resulting from applied cooling.
Acknowledgements
I would like to thank George Ross MD and Tom Ramsey PhD for going out of their way to support this research.
Disclosure
None of the authors identify any conflict of interest.
Conflict of Interest
Adam Sprouse-Blum MD, designed the investigational neck wrap and was the lead investigator. The other authors have no conflict(s) to disclose.
References
1. Arnott J. Practical Illustrations of the Treatment of the Principal Varieties of Headache by the Local Application of Benumbing Cold: With Remarks on the Remedial and Anesthetic Uses of Congelation in Diseases of the Skin and Surgical Operations. London: Churchill; 1849.
2. Ucler S, Coskun O, Inan L, et al. Cold Therapy in Migraine Participants: Open-label, Non-controlled, Pilot Study. Evid Based Complement Alternat Med. 2006 Dec;3(4):489–493. [PMC free article] [PubMed]
3. Robbins LD. Cryotherapy for headache. Headache. 1989;29:598–600. [PubMed]
4. Diamond S, Freitag FG. Cold as an adjunctive therapy for headache. Postgrad Med. 1986;79:305–309.[PubMed]
5. Lance JW. The controlled application of cold and heat by a new device (Migra-lief apparatus) in the treatment of headache. Headache. 1988;28:458–461. [PubMed]
6. Friedman M, Peterson S, Behar C, et al. Intraoral chilling versus oral sumatriptan for acute migraine. Heart Dis. 2001 Nov-Dec;3(6):357–361. [PubMed]
7. Bag B, Karabulut N. Pain-relieving factors in migraine and tension-type headache. Int J Clin Pract. 2005;59:760–763. [PubMed]
8. Zanchin G, Maggioni F, Granella F, et al. Self administered pain-relieving manoeuvres in primary headaches. Cephalalgia. 2001;21:718–726. [PubMed]
9. Abramson DI. Physiologic basis for the use of physical agents in peripheral vascular disorders. Arch Phys Med Rehabil. 1965;46:216–244. [PubMed]
10. Dolan MG, Thornton RM, Fish DR, et al. Effects of cold water immersion on edema formation after blunt injury to the hind limbs of rats. J Athl Train. 1997;32:233–237. [PMC free article] [PubMed]
11. Ho SS, Coel MN, Kagawa R, et al. The effects of ice on blood flow and bone metabolism in knees. Am J Sports Med. 1994;22:537–540. [PubMed]
12. Karunakara RG, Lephart SM, Pincivero DM. Changes in forearm blood flow during single and intermittent cold application. J Orthop Sports Phys Ther. 1999;29:177–180. [PubMed]
13. Kwon HJ, Rhee JG, Song CW, et al. Effects of temperature on blood flow in facial tissues. J Oral Maxillofac Surg. 1986;44:790–793. [PubMed]
14. Lee H, Natsui H, Akimoto T, et al. Effects of cryotherapy after contusion using real-time intravital microscopy. Med Sci Sports Exerc. 2005;37:1093–1098. [PubMed]
15. MacAuley DC. Ice therapy: How good is the evidence? Int J Sports Med. 2001;22:379–384. [PubMed]
16. Swenson C, Sward L, Karlsson J. Cryotherapy in sports medicine. Scand J Med Sci Sports. 1996;6:193–200. [PubMed]
17. Clarke RS, Hellon RF, Lind AR. Vascular reactions of the human forearm to cold. Clin Sci. 1958;17:165–179. [PubMed]
18. Daanen HA, Van de Linde FJ, Romet TT, et al. The effect of body temperature on the hunting response of the middle finger skin temperature. Eur J Appl Physiol Occup Physiol. 1997;76:538–543. [PubMed]
19. Jobe JB, Goldman RF, Beetham WP., Jr Comparison of the hunting reaction in normal and individuals with Raynaud’s disease. Aviat Space Environ Med. 1985;56:568–571. [PubMed]
20. Van den Brande P, De Coninck A, Lievens P. Skin microcirculation responses to severe local cooling. Int J Microcirc Clin Exp. 1997;17:55–60. [PubMed]
21. Knight KL. Cryotherapy in Sports Injury Management. Champaign, IL: Human Kinetics; 1995. p. 68.
22. Deal DN, Tipton J, Rosencrance E, et al. Ice reduces edema. A study of microvascular permeability in rats. J Bone Joint Surg Am. 2002;84-A:1573–1578. [PubMed]
23. Knight KL. Cryotherapy in Sports Injury Management. Champaign, IL: Human Kinetics; 1995. pp. 89–90.
24. De Jong RH, Hershey WN, Wagman IH. Nerve conduction velocity during hypothermia in man. Anesthesiology. 1966;27:805–810. [PubMed]
25. Hocutt JE, Jr, Jaffe R, Rylander CR, et al. Cryotherapy in ankle sprains. Am J Sports Med. 1982;10:316–319. [PubMed]
26. Hubbard TJ, Aronson SL, Denegar CR. Does cryotherapy hasten return to participation? A systematic review. J Athl Train. 2004;39:88–94. [PMC free article] [PubMed]
27. Lee JM, Warren MP, Mason SM. Effects of ice on nerve conduction velocity. Physiotherapy. 1978;64:2–6. [PubMed]
28. Martin SS, Spindler KP, Tarter JW, et al. Cryotherapy: An effective modality for decreasing intraarticular temperature after knee arthroscopy. Am J Sports Med. 2001;29:288–291. [PubMed]
29. Stillwell K, editor. Handbook of Physical Medicine and Rehabilitation. 2nd ed. Philadelphia: W.B. Saunders; 1971. pp. 268–272.
30. Merrick MA. Secondary injury after musculoskeletal trauma: A review and update. J Athl Train. 2002;37:209–217. [PMC free article] [PubMed]
31. Melzack R, Wall PD. Pain mechanisms: a new theory. Science. 1965 Nov 19;150(699):971–979.[PubMed]
32. Zachariassen KE. Hypothermia and cellular physiology. Arctic Med Res. 1991;50(Suppl. 6):13–17.[PubMed]
33. Knight KL. Cryotherapy in Sports Injury Management. Champaign, IL: Human Kinetics; 1995. p. 78.
34. Staats PS, Staats A, Hekmat H. The additive impact of anxiety and a placebo on pain. Pain Med. 2001;2:267–279. [PubMed]
35. Staats P, Hekmat H, Staats A. Suggestion/placebo effects on pain: Negative as well as positive. J Pain Symptom Manage. 1998;15:235–243. [PubMed]
36. Headache Classification Committee of the International Headache Society, author. The International Classification of Headache Disorders. Cephalalgia. (2nd Edition) 2004;24(Suppl. 1):1–160.
37. Hróbjartsson Asbjørn, et al. Canadian Medical Association Journal. 2013. Observer bias in randomized clinical trials with measurement scale outcomes: a systematic review of trials with both blinded and nonblinded assessors. [PMC free article] [PubMed]
